The robust growth in automotive end market electronics is rooted in consumer demand for safer and more functional vehicles. Consequently, the need for highly reliable electronic assemblies, capable of meeting the tightest industry standards,
has never been greater. Kimball Electronics, long a leader in the automotive market, remains committed to advancing creative EMS solutions to accommodate ever-evolving technologies and consumer preferences. As a pioneer in producing braking
and steering electronics, Kimball Electronics has acquired unique market experience and knowledge, positioning it as a premiere EMS player for the exacting demands of Advanced Driver Assistance Systems.
Learn what Kimball Electronics
can do for you.
Our automotive group has continuously delivered on time and on quality to our global Tier 1 customers for over 25 years. We are certified to IATF 16949 requirements and work to manage our supply chain to offer the best value to our customers over the course of the product lifecycle.
The Automotive-dedicated team will provide you with a single point of contact over the course of our partnership to ensure the ease of communication necessary to be flexible with market changes, from NPI to Aftermarket Support.
In every project, our goal is to support our customers' needs with the right mix of services, facility build site options, and technical expertise.
Our global footprint, core competencies, Industry 4.0 experience, and core values ensure lasting relationships and global success.
We are focused, aligned, disciplined, and committed to providing our customers with the highest-reliability and highest-quality in automotive electronics manufacturing. Since 1985, we have been committed to advancing creative EMS solutions to accommodate ever-evolving technologies and consumer preferences rooted in the demand for safer and more functional vehicles in the automotive industry.
May 05, 2020
JASPER, Ind., May 04, 2020 (GLOBE NEWSWIRE) -- Kimball Electronics, Inc. (NASDAQ: KE) today announced financial results for its third quarter ended March 31, 2020.
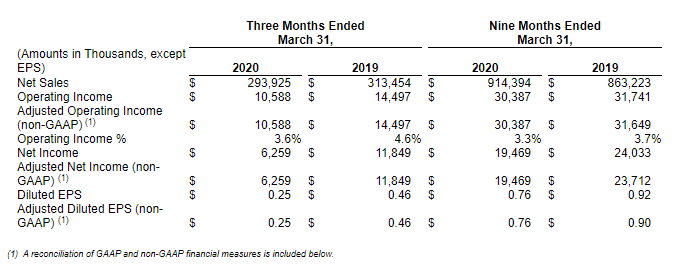
Donald D. Charron, Chairman and Chief Executive Officer, stated, “We are pleased with the results we delivered in the third quarter of fiscal year 2020 despite the interruptions and challenges caused by the COVID-19 pandemic. The safety and health of our employees, customers, suppliers, and our communities are paramount. We are making every effort to keep our facilities safe, following current guidelines suggested by applicable country authorities. Because of the variety of critical medical device assemblies we manufacture around the world, our facilities are classified as “essential businesses” and so all are currently operational, but have been affected to varying degrees by COVID-19.”
Mr. Charron continued, “We serve a diversified portfolio of markets, geographies, and customers. In our third quarter, we experienced a double-digit decline in sales to customers in our medical vertical, which was primarily unrelated to COVID-19. However, we are seeing a significant increase in demand for medical assemblies for the near future, specifically those related to respiratory care and patient monitoring products. We have customers whose products are essential to the health and safety of people around the globe. We are proud of what we do for the world, and we are proud of our people and their extraordinary efforts and contributions during this challenging time. I feel honored and privileged that our Company can play such an important role to help in this pandemic. In our automotive vertical, we started to see the impact of COVID-19 in our third quarter results, although the severity of the impact from the extensive automotive plant shutdowns in North America and Europe will not be reflected in our results until our fiscal fourth quarter.”
Michael K. Sergesketter, Vice President and Chief Financial Officer, stated, “We ended the third quarter with $58.3 million in cash and cash equivalents and debt on our credit facilities of $122.4 million, of which $91.5 million is considered long-term. We had $64.1 million in borrowings available under our credit facilities at March 31, 2020, and we have the ability to increase the borrowing capacity on our primary credit facility by an additional $75 million upon request, subject to consent of the participating lenders, as well as other options to enhance our liquidity. We believe we are in a solid financial position and expect to be able to weather the impact of COVID-19, based on what we know today.”
Third Quarter Fiscal Year 2020 Overview:
.png?sfvrsn=4ac1637d_2)
Forward-Looking Statements
Certain statements contained within this release are considered forward-looking under the Private Securities Litigation Reform Act of 1995 and are subject to risks and uncertainties including, but not limited to, successful integration of acquisitions, ramp up of new operations, global economic conditions, geopolitical environment, global health emergencies including the COVID-19 pandemic, significant volume reductions from key contract customers, loss of key customers or suppliers, financial stability of key customers and suppliers, availability or cost of raw materials, the ability of the supply chain to react successfully to the significant increase in demand for certain medical components, impact related to tariffs and other trade barriers, and increased competitive pricing pressures. Additional cautionary statements regarding other risk factors that could have an effect on the future performance of the Company are contained in its Annual Report on Form 10-K for the year ended June 30, 2019 and other filings with the Securities and Exchange Commission (the “SEC”).
Non-GAAP Financial Measures
This press release contains non-GAAP financial measures. A non-GAAP financial measure is a numerical measure of a company’s financial performance that excludes or includes amounts so as to be different than the most directly comparable measure calculated and presented in accordance with Generally Accepted Accounting Principles (“GAAP”) in the United States in the statement of income, statement of comprehensive income, balance sheet, statement of cash flows, or statement of share owners’ equity of the Company. The non-GAAP financial measures contained herein include adjusted operating income, adjusted net income, adjusted diluted EPS, and ROIC. These measures include adjustments for the nine months ended March 31, 2019 related to proceeds from a class action lawsuit settlement and adjustments to the provision for income taxes resulting from the U.S. Tax Cuts and Jobs Act (“Tax Reform”). Reconciliations of the reported GAAP numbers to these non-GAAP financial measures are included in the financial highlights table below. Management believes it is useful for investors to understand how its core operations performed without the effects of the proceeds from the lawsuit settlement and tax adjustments resulting from Tax Reform. Excluding these amounts allows investors to meaningfully trend, analyze, and benchmark the performance of the Company’s core operations.
| Conference Call / Webcast | |
|---|---|
| Date: | May 5, 2020 |
| Time: | 10:00 AM Eastern Time |
| Live Webcast: | investors.kimballelectronics.com/events-and-presentations |
| Dial-In #: | 800-992-4934 (International Calls - 937-502-2251) |
| Conference ID: | 2576287 |
For those unable to participate in the live webcast, the call will be archived at investors.kimballelectronics.com.
About Kimball Electronics, Inc.
Kimball Electronics is a multifaceted manufacturing solutions provider of electronics and diversified contract manufacturing services to customers around the world. From our operations in the United states, China, India, Japan, Mexico, Poland, Romania, Thailand, and Vietnam, our teams are proud to provide manufacturing services for a variety of industries. Recognized for a reputation of excellence, we are committed to a high-performance culture that values personal and organizational commitment to quality, reliability, value, speed, and ethical behavior. Kimball Electronics, Inc. (NASDAQ: KE) is headquartered in Jasper, Indiana.
To learn more about Kimball Electronics, visit: www.kimballelectronics.com.
Lasting relationships. Global success.
Leave a commentOrder by
Newest on top Oldest on top